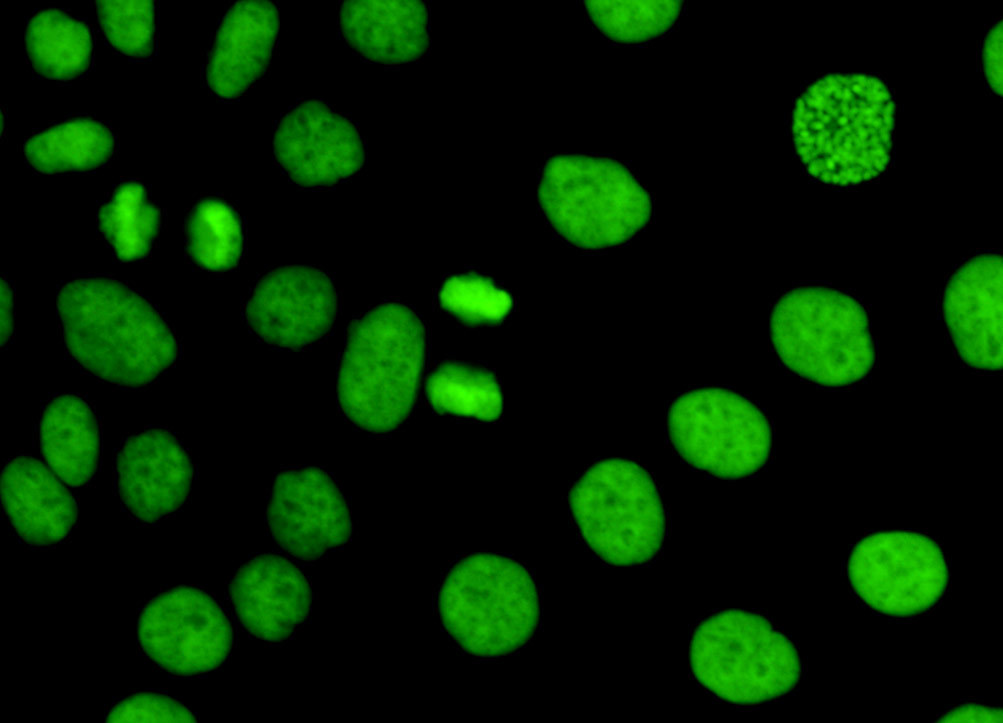
HeLa Cell Histones
B-2E/C Bandpass Emission (Medium Bandwidth Excitation) Blue Set
Fluorescence emission intensity from a culture of HeLa cells that were immunofluorescently labeled with primary anti-histone (pan) mouse monoclonal antibodies followed by goat anti-mouse Fab fragments conjugated to fluorescein. The visible light absorption maximum of fluorescein is 494 nanometers and the emission maximum occurs at 519 nanometers (in the green region of the visible light spectrum). The primary antibody, produced from purified nuclear fractions of HeLa cells, binds specifically to an antigen determinant that is present on all five histone proteins (H1, H2A, H2B, H3, and H4). Note the bright green fluorescence emission exhibited by nuclei in the preparation.















