Swept Field Confocal Microscopy Video Gallery
Live-cell imaging requires increasingly faster instrumentation in order to capture the exceedingly rapid events that occur in subcellular structures. The digital videos presented in this gallery explore several of the most popular fluorescent proteins in fusions with subcellular localization peptides and proteins. Animal and human cell lines expressing fluorescent protein fusions were imaged with a Photometrics Cascade II electron multiplying digital camera (EMCCD) system coupled to a Nikon swept field confocal microscope.
Gray Fox Lung Fibroblast Cells Expressing mEGFP-EB3
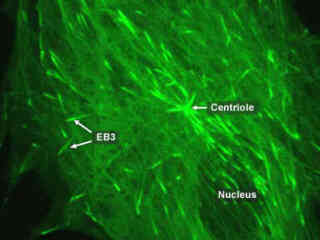
Microtubules are tiny structures that are 25 nanometers in diameter and essential to the cytoskeletal structural network that bestows upon the cell its form and shape. These versatile organelles also act as conveyer belts transporting macromolecular complexes and organelles throughout the cytoplasm. Microtubules change size and shape according to the immediate needs of the cell during its life cycle.
Gray Fox Lung Fibroblast Cells Expressing YPet Fluorescent Protein-EB3
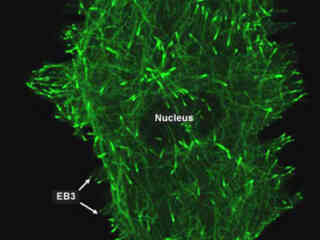
A wide range of microtubule binding proteins, including a family of end-binding proteins (known as EBs), have been demonstrated to associate specifically with the ends of growing microtubules in a variety of cell types. These proteins are believed to regulate microtubule dynamics and the binding of microtubules to organelles, membrane components, and other protein complexes.
Observing Microtubule End-Binding Proteins Fused to mKusabira Orange Fluorescent Protein at 60X Magnification
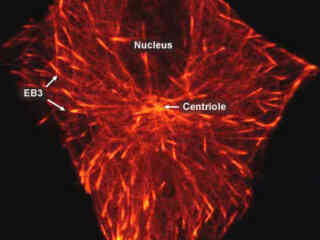
The ends of microtubules are in a constant state of dynamic flux as the constant addition or removal of tubulin proteins alters the overall length of the biopolymer. The rate of depolymerization is different for each end according to its polarity, positive or negative. The fastest growing terminus of a microtubule is termed positive.
Observing Microtubule End-Binding Proteins Fused to mKusabira Orange Fluorescent Protein at 100X Magnification
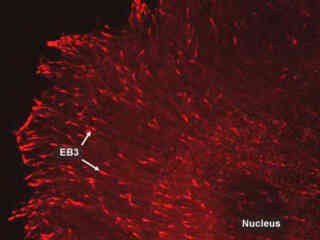
Vesicles, organelles, and a wide spectrum of additional biological macromolecules are ferried throughout the cytoplasm by microtubules. Mitochondria are transported back and forth through the cell by this mechanism, while nerve cells use the ubiquitous microtubule network to translocate vesicles containing neurotransmitters along their axons.
Gray Fox Lung Fibroblast Cells Expressing mEGFP-Tubulin
Microtubules are anchored in the centrosome near the cell center adjacent to the nucleus, but are still able to reach across the cytoplasm and influence events at the cell cortex and plasma membrane. Unlike actin filaments, which usually work in interconnected bundles, microtubules tend to be solitary structures as they provide tracks for other organelles to translate throughout the cytoplasm.
Exploring Tubulin Dynamics in Fibroblast Cells
Tubulin assembly produces microtubules of variable length, but with a precisely determined diameter close to 25 nanometers. The cylindrical walls are about 5 to 6 nanometers thick and the central channel, or lumen, is about 14 nanometers in diameter.
Tubulin Dynamics in Fox Lung Cells
In order to visualize mitosis using standard epi-fluorescence microscopy techniques, fluorescent probes are attached to specific subcellular components, such as the chromosomes and microtubules. The technology has benefited from the use of genetically-encoded fluorescent proteins that can be directly fused to histones and tubulin.
Fox Lung Fibroblast Cells Expressing EGFP fused to Actin
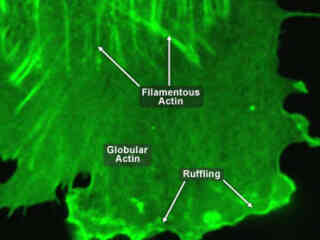
Actin differs by no more than 20 percent in species as diverse as algae and humans. The monomeric subunit of microfilaments, actin participates in many important cellular functions, including muscle contraction, cell motility, cell division and cytokinesis as well as the establishment and maintenance of cell junctions and cell shape.
Actin Dynamics in Epithelial Cells
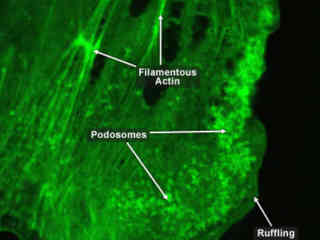
Actin filaments, when combined with other proteins and structures, become stronger and more specialized to perform various tasks within and between cells. This array of filaments and proteins allows them to slide past each other, providing the elasticity necessary for muscle contraction.
Observing EYFP-Actin in Opossum Kidney Cells
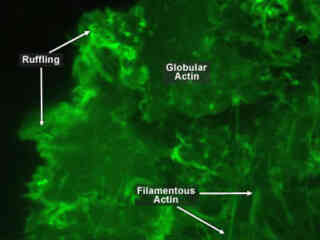
One of the most notable properties of actin is its ability to very quickly assemble into long polymer rods called microfilaments which have a variety of roles. Microfilaments form part of the cell's cytoskeleton, interact with myosin to permit movement of the cell, and pinch the cell into two during cell division.
Imaging of Clathrin Vesicles in Fibroblast Cells
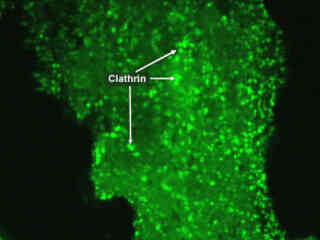
Clathrin molecules are recruited with the aid of adaptor proteins to a membrane segment that is destined to be incorporated into a vesicle. In synaptic vesicle formation, one such adaptor protein is AP180. This protein both recruits clathrin to membranes and also promotes its polymerization on the plasma membrane.
Imaging of Mitochondria with Orange Fluorescent Proteins
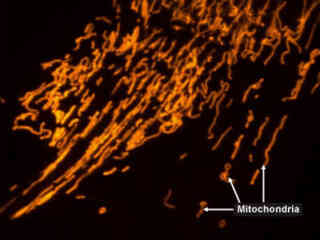
The number of mitochondria present within a cell is directly related to the cell's metabolic needs, and a typical cell can contain anywhere from one to thousands of mitochondria. Complex, multi-celled organisms have evolved only because of the role of mitochondria in their development.
Imaging of Mitochondria with Yellow Fluorescent Proteins
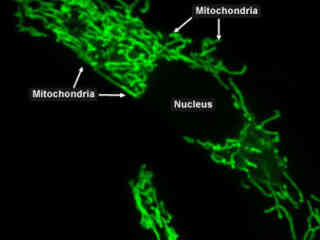
Considered the power generators of the cell, mitochondria convert various nutrients, including oxygen, into adenosine triphosphate (ATP), the chemical "currency" of the structure. These rod-shaped organelles are considered the power generators and drive metabolic activity. The conversion process responsible for the formation of ATP inside mitochondria is an aerobic activity requiring oxygen to proceed.
Peroxisomes Labeled with EGFP
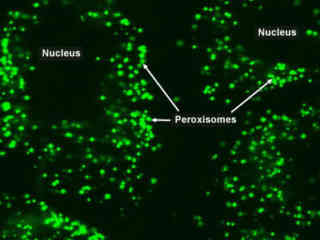
Peroxisomes are relatively small organelles found in eukaryotic cells that are round in shape, and several hundred may occupy a single cell. The peroxisome is contained in a protective membrane of proteins and lipids, and the interior lumen contains oxidation enzymes. It is thought that peroxisomes slowly evolved due to pressures brought about by the increase of oxygen in the environment. Like mitochondria, peroxisomes are self-replicating. Though peroxisomes resemble lysosomes, they have a crystalline structure and are not formed in the Golgi apparatus.
Opossum Kidney Cells Expressing EGFP-Endosomes
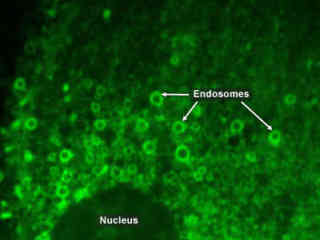
Endosomes are organelles that are formed via a complex cascade of processes collectively known as endocytosis, which involves the invagination (folding inward) of a cell’s plasma membrane to surround macromolecules or other materials diffusing through the extracellular fluid. The encircled foreign materials are then brought into the cell, and following a pinching-off of the membrane (known as budding), are released to the cytoplasm in a sac-like vesicle.
Epithelial Cell Lysosome Dynamics
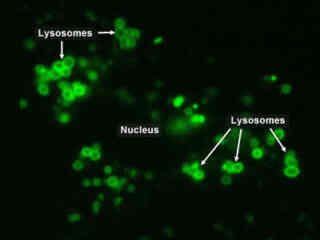
The digestive enzymes that operate in the lysosomes are only active when they reside in an acidic environment. For this reason, the lysosomes contain a very low internal pH range, which ensures that if internal enzymes are accidentally released into the cytoplasm (for example, if the protective lysosome membrane is disrupted), they will not digest functional proteins and lead to cell damage or death.
Golgi Complex Labeled with EGFP
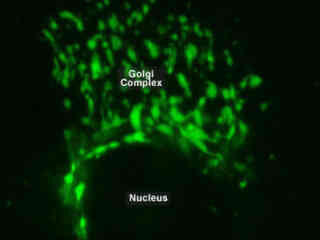
A large and varied group of macromolecules required for normal functon are synthesized within each cell. The Golgi apparatus is integral in modifying, sorting, and packaging these materials for cell secretion (exocytosis) or for use within the cell. The organelle primarily modifies proteins delivered from the rough endoplasmic reticulum but is also involved in the transport of lipids around the cell and the creation of lysosomes.
Golgi Complex Labeled with EYFP
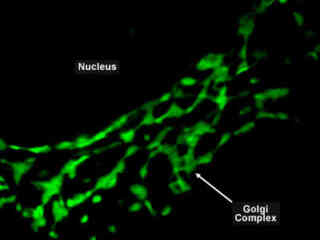
The Golgi complex contains a large quantity of vesicles that are used, in part, to process and send molecules to the cellular membrane where they are excreted. There are also larger secretory vesicles, which are used for selective excretion. The Golgi is principally responsible for directing molecular traffic in the cell as nearly all molecules pass through the Golgi complex at some point in the lifecycle.













