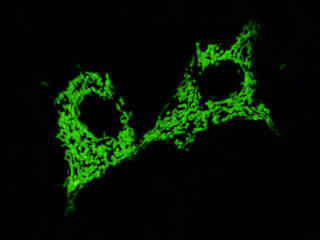
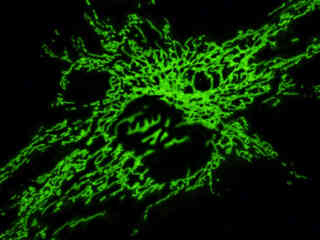
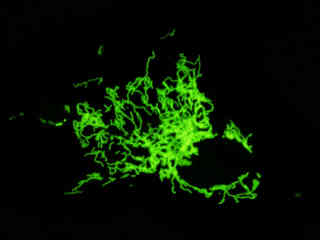
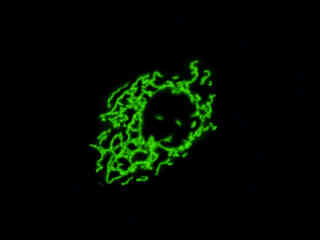
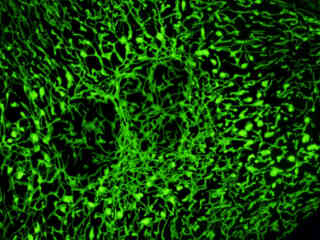
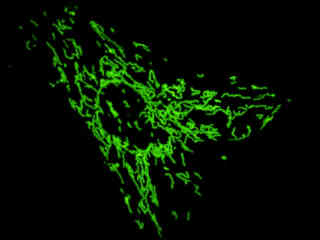
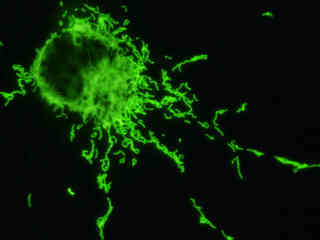
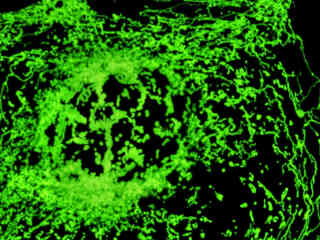
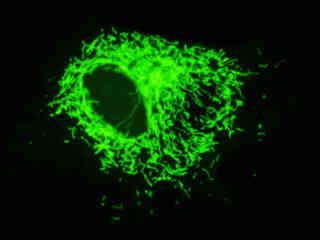
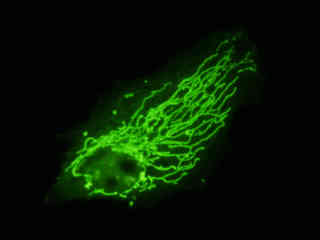
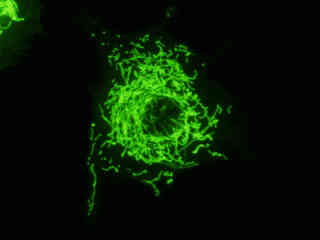
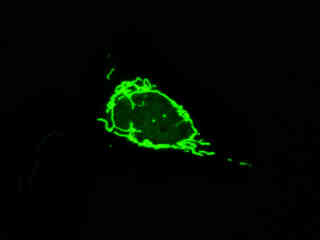
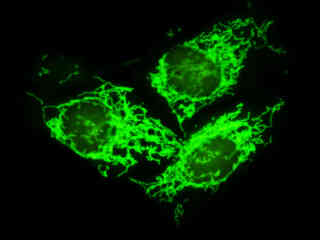
Localization of specific peptides, proteins, and macromolecular complexes to the mitochondria in mammalian cells is generally accomplished through the use of peptide signals that mediate transport to the organelle. Recombinant plasmids have been constructed that contain a fusion protein consisting of the yellow-green variant (referred to as enhanced yellow fluorescent protein; EYFP) of the Aequorea victoria green fluorescent protein (GFP) coupled to the mitochondrial targeting nucleotide sequence from subunit VIII of human cytochrome C oxidase. Upon transcription and translation of the plasmid in transfected mammalian hosts, the mitochondrial localization signal is responsible for transport and distribution of the fluorescent protein chimera throughout the cellular mitochondrial network. Tubular mitochondria can be subsequently visualized using fluorescence microscopy, as illustrated for a variety of established adherent cell lines in Figure 1. The single bandpass emission filter featured by the Nikon YFP HYQ optical block, which was employed to capture these images, produces sharp contrast with little interference from autofluorescence or other fluorescent species.
Plasmid pEYFP-Mitochondria vector gene product expression in various cell types (from both transiently and stably transfected clones; see Figure 1) occurs due to the efficient intracellular translation of a fusion nucleotide sequence combining the enhanced yellow fluorescent protein domain with the mitochondria targeting sequence from subunit VIII of human cytochrome C oxidase, as discussed above. The addition of simian virus 40 (SV40) polyadenylation signals, which are inserted downstream from the chimeric EYFP-Mitochondria fusion sequence, assures proper processing of the transcribed messenger RNA 3' terminus. The fluorescence excitation maximum of EYFP is 513 nanometers and the corresponding emission maximum occurs at 527 nanometers, with a relatively high (approximately 0.60) fluorescence quantum yield. In addition to the four chromophore mutations that shift the fluorescence emission maximum, the nucleotide coding sequence of the EYFP gene contains over 190 silent base alterations, which correspond to human codon-usage preferences that increase translational efficiency.
The collection of specimens illustrated in Figure 1 demonstrates the effectiveness of the Nikon YFP HYQ filter combination for imaging a series of cell lines containing the chimeric EYFP plasmid vector that expresses a fluorescent fusion protein targeted at the intracellular mitochondrial network. The fusion protein is transported into the mitochondria to enable visualization of the subcellular structure in living and fixed cells. Susceptible adherent cell cultures were transfected with the appropriate vector using proprietary lipophilic reagents, and were then cultured for a period of at least 24 hours in nutrient medium supplemented with fetal bovine serum to allow high expression levels of the fluorescent fusion protein.
The enhanced yellow fluorescent protein gene used in these studies contains four amino acid substitutions that shift the emission maximum of green fluorescent protein (GFP) by 18 nanometers, from approximately 509 to 527 nanometers. The gene is optimized with human codons (as described above) and features a consensus Kozak translation initiation signal to achieve higher expression levels in mammalian cell cultures. In general, vectors targeted at specific subcellular organelles contain a fusion gene segment, which couples the EYFP gene to a peptide sequence or complete protein that is localized to a region of interest in living cells.
Additional EYFP-Mitochondria Images with the YFP HYQ Filter Combination
Normal Pig Kidney Cell Mitochondria
Fluorescence emission intensity from a culture of normal pig kidney epithelial (LLC-PK1 line) cells that were transfected with a pEYFP-Mitochondria plasmid subcellular localization vector.
Normal African Green Monkey Kidney Cell Mitochondria
Fluorescence emission intensity from a culture of normal African green monkey kidney fibroblast (CV-1 line) cells that were transfected with a pEYFP-Mitochondria plasmid subcellular localization vector.
Normal Human Fetal Lung Cell Mitochondria
Fluorescence emission intensity from a culture of normal human fetal lung fibroblast (MRC-5 line) cells that were transfected with a pEYFP-Mitochondria plasmid subcellular localization vector.
Human Lung Carcinoma Cell Mitochondria
Fluorescence emission intensity from a culture of human lung carcinoma epithelial (A-549line) cells that were transfected with a pEYFP-Mitochondria plasmid subcellular localization vector.
Normal Canine Kidney Cell Mitochondria
Fluorescence emission intensity from a culture of normal canine kidney epithelial (MDCKline) cells that were transfected with a pEYFP-Mitochondria plasmid subcellular localization vector.
Indian Muntjac Deer Skin Cell Mitochondria
Fluorescence emission intensity from a culture of normal Indian Muntjac deer skin fibroblast cells that were transfected with a pEYFP-Mitochondria plasmid subcellular localization vector.
Albino Swiss Mouse Embryo Cell Mitochondria
Examine the fluorescence emission intensity from a culture of albino Swiss mouse embryo fibroblast (3T3 line) cells that were transfected with a pEYFP-Mitochondria plasmid subcellular localization vector. Note the relatively constant level of green fluorescence defining the tubular mitochondrial geometry, which exists as an extended network traversing the cytoplasm. In many cases, mitochondria are concentrated near the nucleus, leading to an increase in fluorescence emission from this region.
Transformed African Green Monkey Kidney Cell Mitochondria
A culture of simian virus 40 (SV40) transformed African green monkey kidney fibroblast (COS-7 line) cells were transfected with a pEYFP-Mitochondria plasmid subcellular localization vector, and subsequently cultured in nutrient medium for a minimum of 24 hours before recording images.
Rat Thoracic Aorta Myoblast Cell Mitochondria
This section reviews fluorescence emission intensity observed with the Nikon YFP HYQ blue-green excitation filter combination in a culture of rat thoracic aorta myoblast (A-10 line) cells labeled with a chimera of enhanced yellow fluorescent protein (EYFP) and the mitochondrial targeting sequence from subunit VIII of human cytochrome c oxidase.
Bovine Pulmonary Artery Endothelial Cell Mitochondria
Observe the fluorescence emission intensity from a culture of bovine pulmonary artery endothelial (BPAE line) cells that were transfected with a pEYFP-Mitochondria plasmid subcellular localization vector. Note the relatively constant level of green fluorescence defining the tubular mitochondrial geometry, which exists as an extended network traversing the length of the cytoplasm.
Rat Enteroglial Cell Mitochondria
Rat jejunum myenteric plexus enteroglial (EGCline) cells were transfected with a pEYFP-Mitochondria plasmid subcellular localization vector to reveal the distribution of this organelle within the cytoplasm. As is evident from the image in this section, the tubular mitochondria surround the nucleus and extend to the periphery of the cytoplasm in this neural cell line.
Human Osteosarcoma Bone Cell Mitochondria
The mitochondria in this cell line (U2OS) appear as elongated tubules that surround the nucleus, but often do not spread throughout the cytoplasm as they do in other cells.
Human Cervical Carcinoma Cell Mitochondria
Review the fluorescence emission intensity from a culture of human cervical carcinoma epithelial (HeLa line) cells that were transfected with a pEYFP-Mitochondria plasmid subcellular localization vector, and then cultured in nutrient medium for a minimum of 24 hours before recording images.